July 11, 2018
Massey becomes first treatment center in Virginia to offer FDA-approved CAR T-cell therapy
Share this story
VCU Massey Cancer Center is now an official treatment center for Yescarta™ by Kite Pharma for adult patients living with certain types of non-Hodgkin’s lymphoma (recurrent or refractory B-cell lymphoma), making it the first treatment center in Virginia to offer an FDA-approved CAR T-cell therapy. Massey, in partnership with Children’s Hospital of Richmond at VCU, also will soon be offering another FDA-approved CAR T-cell therapy, KYMRIAH™ by Novartis, for children and young adults with recurrent or refractory B-cell acute lymphoblastic leukemia.
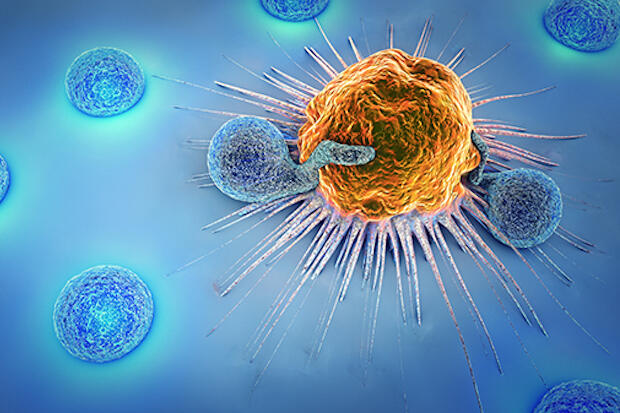
CAR T-cell therapy is a new type of immunotherapy called adoptive cell therapy in which a patient’s T-cells, a type of immune system cell, are genetically modified so they will attack cancer cells. T-cells are taken from a patient’s blood through a process called apheresis, in which blood is collected from the vein, the T-cells are separated from the blood by a machine and then the remaining blood is returned to the patient. The T-cells are then genetically modified in a laboratory before millions of copies are given back to the patient by infusion.

“We’re excited to offer these promising new immunotherapies for patients throughout the mid-Atlantic region,” said John M. McCarty, M.D., a professor in the VCU School of Medicine and director of Massey’s Cellular Immunotherapies and Transplantation Program. “We are continuously working to expand the number of cellular immunotherapies we offer for both adult and pediatric patients, and we plan to bring on additional FDA-approved therapies as well as clinical trials as early as this fall.”
Leading this effort is Massey’s Bone Marrow Transplantation Program, which has been renamed as the Cellular Immunotherapies and Transplantation Program to reflect the expanded therapeutic options that now include bone marrow and stem cell transplantation as well as the most recent cellular immunotherapies available.
CAR T-cell therapy has proven effective at treating advanced blood cancers in children and adults. These cancers are usually treated successfully with chemotherapy, but in some cases conventional approaches do not work. That is when CAR-T cell therapy can be the best option and may even allow some patients to avoid a bone marrow transplant.
A major advantage of CAR T-cell therapy is that it is a single infusion that usually requires one week of inpatient care, as opposed to the six months or more of chemotherapy, specialized post-transplant care and lifestyle restrictions typically required for patients with non-Hodgkin’s lymphoma and childhood leukemia patients no longer responding to conventional therapies.
Also, the benefits of CAR T-cell therapy may last for years. Since the cells can persist in the body long term, the patient’s own immune system is effectively reprogrammed to recognize and attack cancer cells to prevent relapse.
Several patients have already undergone consultation for treatment. McCarty said that each patient is evaluated before the team recommends the combination and sequence of treatments they believe will provide the best possible outcomes and quality of life. For some, CAR T-cell therapy may be the primary treatment while others may benefit more from stem cell transplantation or a combination of the two approaches.

“We extensively evaluate each patient and work with their referring physicians to create a personalized treatment plan that carefully weighs the risks and benefits of each approach,” said Gary Simmons, D.O., assistant professor in the School of Medicine and deputy director of Massey’s Cellular Immunotherapies and Transplantation Program. “While CAR T-cell therapies can potentially offer lasting and durable remission from cancer, they also can carry a risk for serious side effects related to how they work.”
Effectively managing the potential side effects requires collaboration among many different specialties, which is why these gene therapies are typically only found at National Cancer Institute-designated centers, such as Massey, and large academic hospitals. The most serious side effect is cytokine release syndrome, also known as a “cytokine storm,” which is an inflammatory response caused by the immune system. The symptoms may appear benign at first, but can rapidly worsen and lead to neurological complications. This process of cytokine hyperactivity is actually a necessary part of expanding the activity and longevity of the CAR T-cells, so having a team that can modulate the impact of this process while supporting the patient is a key component to the therapies’ success.
“Massey’s strong neuro-oncology team and long-standing clinical collaborations with our critical care team and pediatric experts at Children's Hospital of Richmond at VCU are key parts of our comprehensive program and make us an ideal site for this novel treatment,” McCarty said. “Patients receiving CAR T-cell therapy at Massey will stay at the hospital for one week after infusion, and they must remain within a 30-minute drive of Massey for an additional month in order for the care team to be able to rapidly and effectively address the unique side effects that can occur with this therapy. During that time, they are advised to visit us at the onset of even the mildest symptoms to receive immediate access to care.”
Due to the complexities of CAR T-cell therapies and stem cell transplantation, Massey’s cellular immunotherapy and transplant coordinators guide patients and their caregivers through the entire process. They help coordinate medical records, schedule tests, navigate insurance, assist with transportation, arrange treatment and follow-up appointments, manage side effects, develop survivorship plans and more. VCU Massey’s distinction as a center for complex and rare cancers allows different therapeutic disciplines to contribute to the care of these patients and improve their experience with this potent and novel therapy.
For more information about CAR T-cell therapies or other treatments available through Massey’s Cellular Immunotherapies and Transplantation Program, please visit www.MasseyCIT.org, call (804) 628-2079 or email judith.davis@vcuhealth.org.
Subscribe to VCU News
Subscribe to VCU News at newsletter.vcu.edu and receive a selection of stories, videos, photos, news clips and event listings in your inbox.